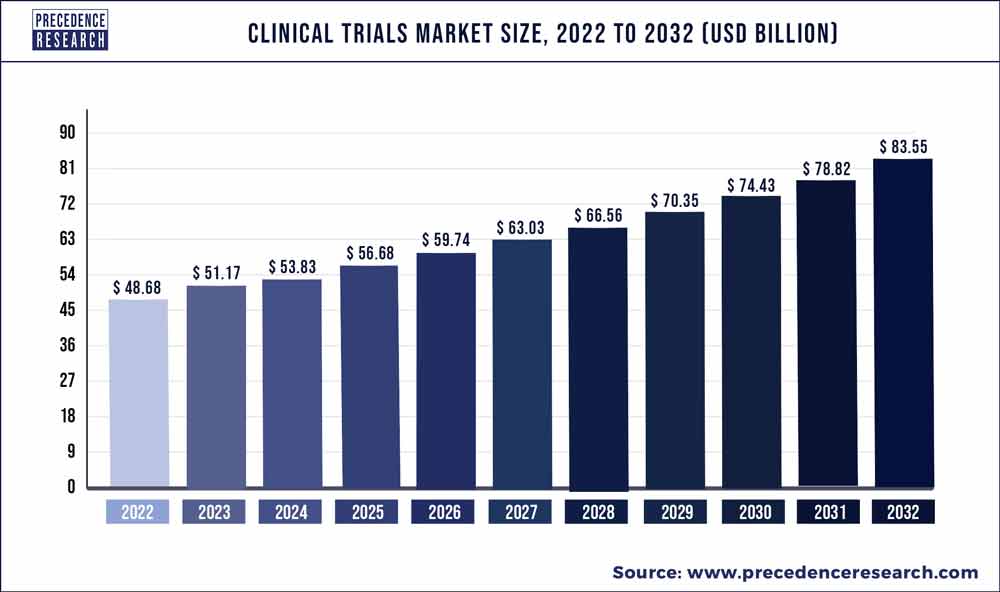
The global clinical trials market size reached USD 48.68 billion in 2022 and is predicted to surpass USD 83.55 bn by 2032 with a registered CAGR of 5.6% from 2023 to 2032.
Key Takeaway:
- North America has held 51.7% of the total market share in 2022.
- Asia Pacific region is growing at a CAGR of 6.8% during the forecast period.
- By indication, the oncology segment has held a market share of around 24.7% in 2022.
- The cardiovascular condition segment is growing at a CAGR of 6.4% during the forecast period.
- By sponsor, The pharmaceutical & biopharmaceutical companies has generated the largest revenue share of around 72% in 2022.
- By Indication by study design, the interventional trials market for autoimmune/inflammation has captured 83% market share in 2022.
- By study design, the interventional design segment has accounted revenue share of 46% in 2022.
- The expanded access trials segment is projected to register at a CAGR of 6.4% over the forecast period.
The clinical trials market is expanding because to the rising frequency of chronic disorders and the rising demand for clinical trials in developing nations. The clinical trials market is fueled by the increasing number of biologics on the market and the demand for contract research organizations to undertake clinical studies. In addition, various biotechnology and pharmaceutical companies are now conducting clinical trials for severe chronic and infectious disorders such as HIV and cancer, which will help to expand the clinical trials market.
Get the Report Sample Copy@https://www.precedenceresearch.com/sample/1185
Regional Snapshot
Asia-Pacific is the largest segment for clinical trials market in terms of region. This is attributed to the growing availability of a broad patient pool, making candidate recruiting easier. The global COVID-19 pandemic is also a major contributor to the clinical trials market expansion.
North America region is the fastest growing region in the clinical trials market. This can be linked to the North America region’s increased research and development adoption of new clinical research technology. Furthermore, favorable government assistance for clinical trials in the U.S. is expected to stimulate demand.
Report Highlights
- Based on the phase, the phase III segment dominated the global clinical trials market in 2020 with highest market share. The fact that a phase III study covers a high number of individuals and is the most expensive phase of a trial among all segments contributes to the clinical trials market growth.
- Based on the study design, the interventional design segment dominated the global clinical trials market in 2020 with highest market share. Interventional studies account largest percentage of all investigations, with the bulk of them using medicines or biologics, followed by clinical procedure, behavioral, and device intervention studies.
- Based on the indication, the oncology segment dominated the global clinical trials market in 2020 with highest market share. The oncology is the study of tumors. Tumors have the potential to be life-threatening in a large number of individuals, necessitating the development of better and more advanced treatments for diverse forms of tumors.
Report Scope of the Clinical Trials Market
| Report Highlights | Details |
| Market Size | USD 83.55 Billion by 2032 |
| Growth Rate | CAGR of 5.6% from 2023 to 2032 |
| Largest Market | North America |
| Fastest Growing Market | Asia Pacific |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Segments Covered | Phase, Study Design, Indication |
Clinical Trials Market Dynamics
Drivers
Surge in demand for outsourcing clinical trials
The demand for efficient, fast-paced, and trustworthy clinical trials programs is expected to expand as the demand for novel medications and improved medical technologies grows. Furthermore, the medication development process is exceedingly dangerous for biotechnology and pharmaceutical businesses, with much lower approval rates and accompanying expansive expenses. As a result, outsourcing the clinical trials program to several contract research organizations (CRO) has been determined to save the pharmaceutical corporation substantial time and money. Regional penetration of specific contract research organizations (CRO) has also been documented. Thus, the surge in the demand for outsourcing clinical trials is propelling the growth of the clinical trials market growth during the forecast period.
Restraints
High cost of clinical trials
The clinical trials market’s services are costly. In the forecast period, market labor costs are a restricting factor for the market growth. Patenting and contracting for the clinical trials market is a complicated process. As a result, the clinical trials market’s labor costs are high. The cost is an issue since it reduces demand in a few markets. The clinical trials services must be cost-effective in most businesses. The high cost, on the other hand, raises the industry’s overall operating costs. Thus, the high cost of clinical trials is hindering the growth of the clinical trials market during the forecast period.
Opportunities
Growing use of predictive analytics
Several firms are already using predictive analytics techniques such as artificial intelligence and machine learning to construct models and advise choices. Given the wealth of health data now available to clinical trial investigators, predictive analytics tools can be used in clinical trial design to identify patient characteristics that are more likely to respond to a specific treatment pattern, thereby increasing success rates and lowering risk in large, multi-center clinical trials. As a result, the growing use of predictive analysis is creating lucrative opportunities for the market growth during the forecast period.
Challenges
Stringent government regulations
Conducting clinical trials in different nations comes with a slew of regulatory challenges that could stymie market growth. The restrictions can be simplified with the cooperation of multiple regulatory authorities, however there is now a slow operating speed. Several nations also demand local language translation, as well as import and export authorization and the presentation of data on local patients. As a result, a complicated regulatory structure complicated regulatory structure combined with a considerable language barrier could stifle regional development.
Read Also: Non-Invasive Aesthetic Treatment Market Size, Report By 2033
Top Players contending in the Market:
- Parexel
- IQVIA
- Charles River Laboratory
- Omnicare
- Kendle
- Chiltern
- Pharmaceutical Product Development, LLC
Major Market Segments Covered:
By Phase
- Phase 1
- Phase 2
- Phase 3
- Phase 4
By Study Design
- Observational
- Interventional
- Expanded Access
By Indication
- Autoimmune/Inflammation
- Rheumatoid arthritis
- Multiple Sclerosis
- Osteoarthritis
- Irritable Bowel Syndrome (IBS)
- Others
- Pain Management
- Chronic Pain
- Acute Pain
- Oncology
- Blood Cancer
- Solid Tumors
- Other
- CNS Condition
- Epilepsy
- Parkinson’s Disease (PD)
- Huntington’s Disease
- Stroke
- Traumatic Brain Injury (TBI)
- Amyotrophic Lateral Sclerosis (ALS)
- Muscle Regeneration
- Others
- Diabetes
- Obesity
- Cardiovascular
- Others
By Service Type
- Protocol Designing
- Patient Recruitment
- Laboratory Services
- Site Identification
- Bioanalytical Testing Services
- Cell-based Assays
- Virology Testing
- Method Development, Optimization, & Validation
- Serology, Immunogenicity, & Neutralizing Antibodies
- Biomarker Testing Services
- PK/PD (Pharmacokinetics/Pharmacodynamics) Testing Services
- Other Bioanalytical Testing Services
- Analytical Testing Services
- Clinical Trial Supply & Logistic Services
- Clinical Trial Data Management Services
- Decentralized Clinical Services
- Medical Device Testing Services
- Others
By Sponsor
- Pharmaceutical & Biopharmaceutical Companies
- Medical Device Companies
- Others
By End User
- Hospital
- Laboratories
- Clinics
By Application
- Vaccine
- Cell & Gene Therapy
- Small Molecules
- Other Applications
By Geography
-
- North America
- U.S.
- Canada
- Europe
- Germany
- France
- United Kingdom
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Rest of Latin America
- Middle East & Africa (MEA)
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa
- North America
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/
- Esoteric Testing Market Size to Hit USD 75.54 Bn by 2032 - July 17, 2024
- mRNA Therapeutics Market Size to Surpass USD 39.99 Bn By 2033 - July 17, 2024
- Acne Treatment Market Size to Hit USD 15.86 Bn by 2033 - July 17, 2024