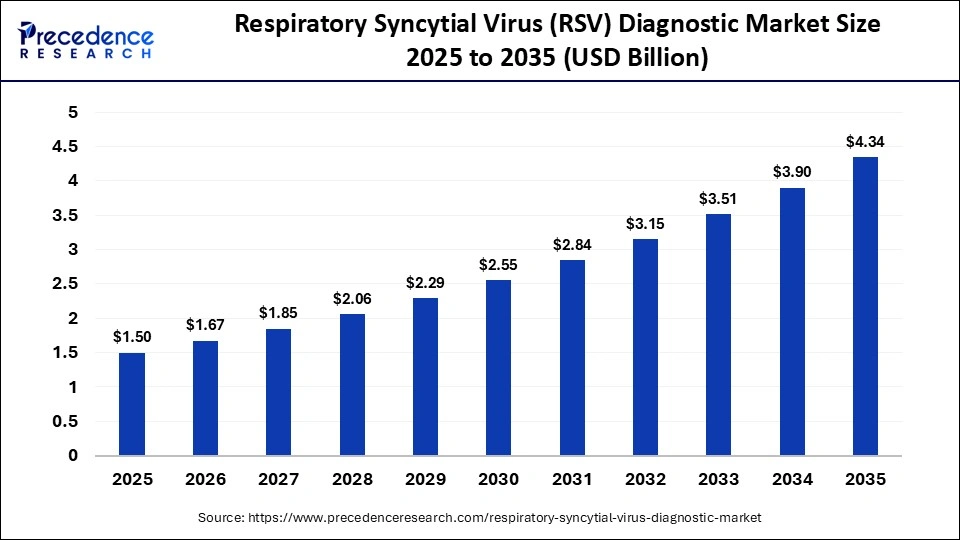
The global respiratory syncytial virus diagnostic market is witnessing strong growth due to the increasing burden of respiratory infections and rising demand for early and accurate detection. The market was valued at USD 1.50 billion in 2025 and is expected to grow from USD 1.67 billion in 2026 to USD 4.34 billion by 2035, expanding at a CAGR of 11.20%.
RSV is a highly contagious virus that primarily affects infants, elderly individuals, and immunocompromised populations. With seasonal outbreaks and rising hospitalization rates, the importance of rapid and reliable diagnostic tools has become more critical than ever.
Read Also: Cholestatic Pruritus Market
What is RSV and Why is Diagnosis Important?
Respiratory syncytial virus (RSV) is a leading cause of lower respiratory tract infections, particularly in young children and older adults. Early diagnosis is essential for:
- Timely clinical intervention
- Preventing severe complications
- Reducing hospital admissions
- Controlling outbreaks
Modern RSV diagnostics include molecular testing, rapid antigen detection, and point-of-care solutions, enabling faster and more accurate results.
Respiratory Syncytial Virus Diagnostic Market Dynamics
Key Growth Drivers
1. Rising Prevalence of Respiratory Infections
The increasing incidence of RSV infections globally is a major factor driving market growth. Seasonal outbreaks and growing awareness are boosting testing demand.
2. Growing Neonatal and Geriatric Population
Infants and elderly individuals are highly vulnerable to RSV, leading to increased diagnostic demand in hospitals and clinics.
3. Expansion of Point-of-Care Testing
The shift toward decentralized healthcare has increased adoption of rapid and portable diagnostic tools, enabling faster decision-making in emergency settings.
4. Increasing Vaccination and Preventive Therapies
With the rise of RSV vaccines and monoclonal antibody treatments, diagnostics are becoming essential for screening and treatment eligibility.
Role of AI in the RSV Diagnostic Market
Artificial intelligence is transforming RSV diagnostics by enabling predictive and data-driven healthcare solutions.
AI algorithms are increasingly used to analyze epidemiological trends, weather patterns, and hospital data to predict RSV outbreaks before they occur, allowing healthcare systems to prepare in advance.
Additionally, AI enhances diagnostic accuracy by improving the interpretation of multiplex test results, especially when RSV symptoms overlap with influenza or COVID-19. This supports clinicians in making faster and more accurate decisions.
Key Market Trends
Shift Toward Multiplex Testing
Healthcare providers are increasingly adopting multiplex diagnostic panels that can detect RSV along with other respiratory pathogens in a single test.
Rise of Point-of-Care Molecular Platforms
Compact molecular diagnostic devices are enabling PCR-level accuracy outside traditional labs, improving accessibility and speed.
Integration of Real-Time Surveillance Systems
Diagnostic tools are becoming part of connected healthcare ecosystems, enabling real-time disease monitoring and faster public health responses.
Pediatric-Focused Innovation
New diagnostic solutions are being designed specifically for infants, with less invasive sampling techniques and faster turnaround times.
Market Segmentation Analysis
By Product & Service
- Kits and assays (reagents) dominate the market due to consistent demand for consumables in testing.
- Software and services are expected to grow rapidly due to increasing reliance on data analytics and reporting systems.
- Instruments/analyzers remain essential but grow at a slower pace due to high initial costs.
By Technology
- Molecular diagnostics lead the market with a 55% share, offering high sensitivity and accuracy.
- Rapid antigen tests account for 27% share, valued for speed and ease of use.
- Immunoassays continue to support routine laboratory testing.
By End-User
- Hospitals and clinics dominate with a 43.1% share, driven by high patient volumes and need for immediate diagnosis.
- Diagnostic laboratories hold 31.5% share, benefiting from outsourcing trends.
- Point-of-care testing centers are rapidly growing due to decentralized healthcare models.
Regional Analysis
North America
North America leads the market with a 39.2% share, supported by:
- Advanced diagnostic infrastructure
- Strong surveillance programs
- High adoption of molecular technologies
Asia Pacific
Asia Pacific is the fastest-growing region, driven by:
- Expanding healthcare infrastructure
- Increasing awareness of respiratory diseases
- Rising adoption of molecular diagnostics
Europe
Europe is witnessing steady growth due to:
- Government initiatives for infectious disease control
- Strong regulatory support
- Increasing adoption of advanced diagnostic tools
Competitive Landscape
The RSV diagnostic market is highly competitive, with major players focusing on innovation, partnerships, and product development.
Key Companies
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- Becton, Dickinson and Company
- Danaher Corporation (Cepheid)
- Thermo Fisher Scientific Inc.
- bioMérieux SA
- QIAGEN N.V.
- Siemens Healthineers
- Bio-Rad Laboratories
- DiaSorin S.p.A.
Challenges in the Market
Despite strong growth, the market faces several challenges:
- High cost of molecular diagnostic platforms
- Limited access to advanced technologies in developing regions
- Regulatory concerns around at-home testing accuracy
- Need for skilled professionals to operate advanced systems
Get a Sample Copy: https://www.precedenceresearch.com/sample/8321
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
- Respiratory Syncytial Virus Diagnostic Market Size to Reach USD 4.34 Billion by 2035 - April 23, 2026
- Cholestatic Pruritus Market Size to Hit USD 2.55 Billion by 2035 - April 23, 2026
- Amylin Analog Drugs Market to Reach USD 7.10 Billion by 2035 - April 22, 2026