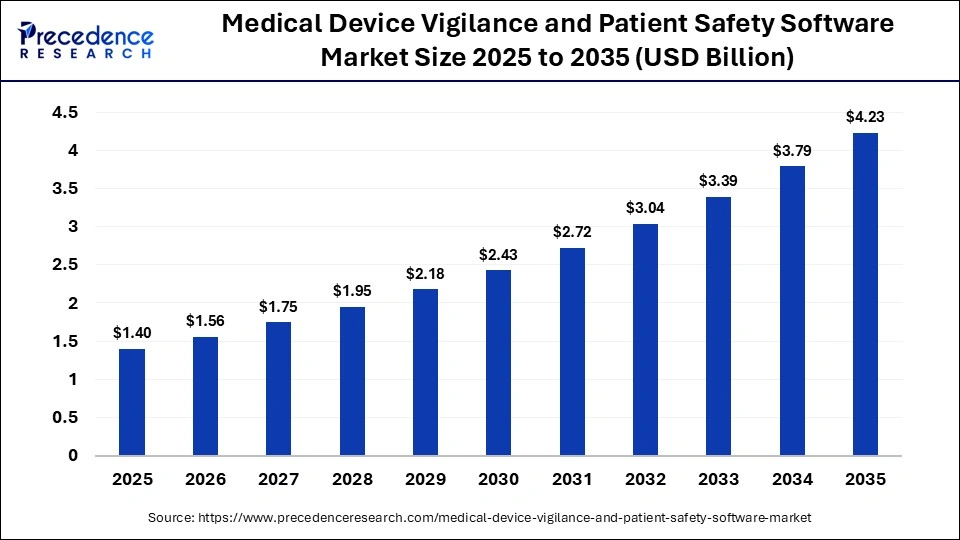
The global medical device vigilance and patient safety software market is witnessing strong growth as healthcare systems increasingly prioritize safety, compliance, and real-time monitoring. The market size was valued at USD 1.40 billion in 2025 and is projected to reach approximately USD 4.23 billion by 2035, growing at a CAGR of 11.70% during the forecast period.
The growing complexity of medical devices, rising regulatory scrutiny, and increasing demand for digital health solutions are driving the adoption of advanced vigilance software. These platforms play a critical role in monitoring adverse events, improving patient outcomes, and ensuring compliance with global healthcare standards.
Read Also: Inflammatory Disease Market
Quick Insights
The market generated USD 1.40 billion in 2025 and is expected to reach USD 4.23 billion by 2035, expanding at a CAGR of 11.70%. North America dominated the market with around 40% share in 2025, while Asia Pacific is projected to grow at the fastest CAGR of 16%. Adverse event reporting software led the segment with a 35% share, and cloud-based deployment dominated with 55% share. Medical device manufacturers accounted for the largest end-user segment with approximately 45% share.
How is Artificial Intelligence Transforming Patient Safety Software?
Artificial Intelligence (AI) is playing a transformative role in medical device vigilance and patient safety software by enabling predictive analytics and automated risk detection. AI-powered systems can identify adverse events, predict medication errors, and assess patient risks such as falls or complications, significantly improving clinical outcomes.
Additionally, AI enhances regulatory reporting and data analysis by leveraging machine learning and neural technologies. These capabilities allow healthcare providers to process large datasets efficiently, detect patterns, and implement proactive safety measures, making patient safety systems more intelligent and responsive.
What are the Key Growth Drivers of the Market?
The market is primarily driven by increasing regulatory requirements and the growing focus on patient safety across healthcare systems. Organizations are adopting advanced software solutions to ensure compliance, reduce risks, and improve transparency in medical device monitoring.
Another major driver is the rising adoption of digital health technologies and real-time monitoring systems. Healthcare providers and manufacturers are investing in software platforms that enable efficient reporting, risk management, and post-market surveillance of medical devices.
What Opportunities and Trends are Shaping the Market?
Is Cloud-Based Deployment Driving Market Expansion?
Yes, cloud-based software is dominating the market due to its scalability, flexibility, and real-time data access capabilities. These platforms enable centralized reporting and seamless integration with healthcare IT systems, improving operational efficiency.
Are AI and Predictive Analytics Creating New Opportunities?
Absolutely. The integration of AI and machine learning is enabling predictive risk analysis and early detection of safety issues, transforming traditional vigilance systems into proactive safety solutions.
Is Cybersecurity Becoming a Key Focus Area?
Yes, with increasing digitalization, healthcare organizations are prioritizing cybersecurity to protect sensitive patient data and ensure the integrity of vigilance systems.
Regional Analysis
North America led the global market in 2025, driven by advanced healthcare infrastructure, strong regulatory frameworks, and widespread adoption of digital safety solutions.
Europe remains a significant market due to stringent regulatory standards and increasing adoption of patient safety technologies. Meanwhile, Asia Pacific is expected to witness the fastest growth, supported by rising healthcare investments, expanding medical device adoption, and growing awareness of patient safety in emerging economies.
Segmental Analysis
By Software Type
| Software Type | 2025 Share | Growth Trend |
|---|---|---|
| Adverse Event Reporting Software | 35% | Dominant |
| Risk Management Software | 25% | Fastest Growing |
| Regulatory Compliance Software | 15% | Steady Growth |
| Incident Management Software | 10% | Moderate Growth |
| Post-Market Surveillance Software | 10% | Stable |
Adverse event reporting software dominated the market due to the increasing need for compliance and safety monitoring, while risk management software is expected to grow rapidly due to proactive risk mitigation strategies.
By Deployment Mode
| Deployment Mode | 2025 Share |
|---|---|
| Cloud-Based | 55% |
| On-Premise | 25% |
| Hybrid | 20% |
Cloud-based solutions lead due to scalability, ease of access, and real-time monitoring capabilities.
By End-User
| End User | 2025 Share |
|---|---|
| Medical Device Manufacturers | 45% |
| Contract Research Organizations | 20% |
| Hospitals & Healthcare Providers | 15% |
| Regulatory Authorities | 10% |
| Others | 10% |
Medical device manufacturers dominate due to the need for compliance and post-market surveillance systems.
Competitive Landscape and Key Companies
The market is highly competitive, with leading players focusing on innovation, partnerships, and AI-driven safety solutions. Key companies include:
- Medtronic plc
- Oracle Corporation
- Veeva Systems Inc.
- IQVIA Inc.
- Siemens Healthineers
- GE Healthcare
- Stryker Corporation
- Wolters Kluwer N.V.
These companies are investing in AI-enabled platforms, cloud-based solutions, and advanced analytics to strengthen their market presence.
What Challenges are Impacting Market Growth?
Are High Costs and Complexity Limiting Adoption?
Yes, high implementation costs and complex software management systems can hinder adoption, especially among smaller healthcare providers. Frequent updates and maintenance requirements add to operational challenges.
Is Data Integration a Challenge?
Integrating vigilance software with existing healthcare systems can be complex, requiring skilled professionals and advanced infrastructure.
Case Study: AI-Driven Patient Safety in Modern Hospitals
Hospitals adopting AI-powered vigilance systems have significantly improved adverse event detection and reporting efficiency. By automating risk identification and enabling real-time monitoring, these systems have reduced medical errors and enhanced patient outcomes, demonstrating the value of digital safety platforms.
Conclusion
The medical device vigilance and patient safety software market is becoming a critical component of modern healthcare systems. With increasing regulatory pressure, technological advancements, and a growing focus on patient safety, the market is poised for sustained growth over the next decade.
Get Sample Copy: https://www.precedenceresearch.com/sample/8345
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com