Infectious Disease Immunoassay Market Size 2026: Growth, Trends, and Forecast to 2035
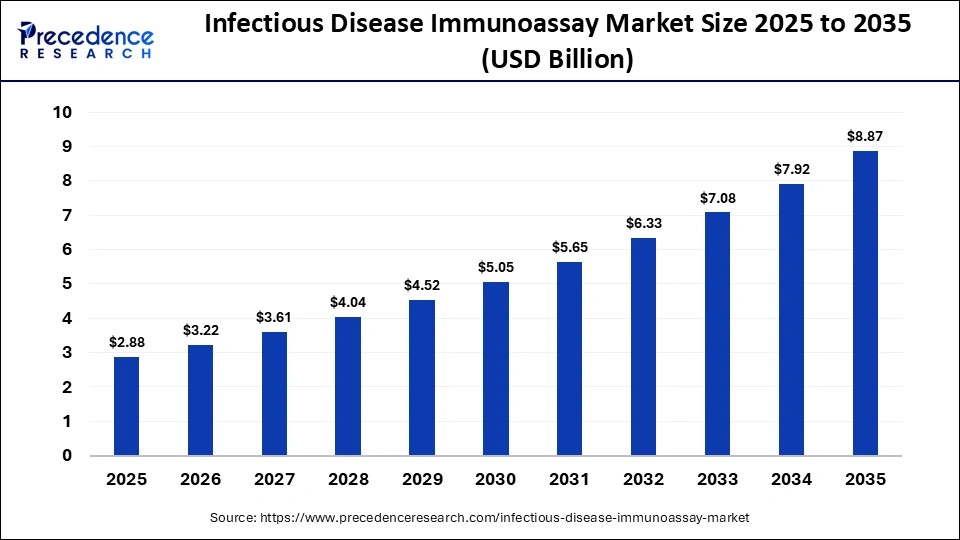
The global infectious disease immunoassay market is witnessing significant growth, driven by the rising burden of infectious diseases and the increasing need for rapid and accurate diagnostic solutions. The market was valued at USD 2.88 billion in 2025 and is expected to grow to USD 3.22 billion in 2026, reaching approximately USD 8.87 billion by 2035, expanding at a CAGR of 11.90% from 2026 to 2035.
The expansion of diagnostic centers, increasing awareness about early disease detection, and advancements in immunoassay technologies are key factors accelerating market growth.
Read Also: Oxygen Therapy Equipment Market
Key Takeaways
- North America dominated the market with a 34.8% share in 2025
- Asia-Pacific is expected to grow at the fastest CAGR during the forecast period
- Reagents & Kits segment led with 56.8% market share in 2025
- Software & Data Management Solutions expected to grow fastest (CAGR 12.3%)
- Chemiluminescent Immunoassay (CLIA) dominated technology segment (34.6%)
- Multiplex Immunoassay to grow at highest CAGR (13.4%)
- Antibody Detection Tests held dominant position in 2025
- Hepatitis Immunoassays led disease segment (23.8%)
- Diagnostic Testing dominated application segment (38.7%)
What is the Infectious Disease Immunoassay Market?
The infectious disease immunoassay market includes reagents, kits, analyzers, calibrators, controls, and software solutions used to detect antigens, antibodies, and immune markers linked to infections caused by viruses, bacteria, fungi, and parasites.
These assays are widely used across:
- Hospitals
- Diagnostic laboratories
- Blood banks
- Clinics
- Public health institutions
Common immunoassay technologies include:
- ELISA (Enzyme-Linked Immunosorbent Assay)
- CLIA (Chemiluminescent Immunoassay)
- LFIA (Lateral Flow Immunoassay)
- RIA (Radioimmunoassay)
- Rapid antigen and antibody tests
Among these, lateral flow immunoassays (LFIA) are highly popular due to their speed, cost-effectiveness, portability, and ease of use, especially in point-of-care settings.
How is AI Transforming the Infectious Disease Immunoassay Market?
Artificial Intelligence (AI) is revolutionizing infectious disease diagnostics by enhancing:
- Accuracy and speed of disease detection
- Pattern recognition in complex biological data
- Automated image analysis in microbiology and radiology
- Pathogen surveillance and outbreak prediction
AI-driven technologies support:
- High-throughput sequencing
- Variant tracking
- Early outbreak detection
- Point-of-care diagnostic improvements
This integration is enabling faster clinical decisions and improved patient outcomes, especially in resource-limited settings.
Key Market Trends
1. Adoption of Advanced In Vitro Models
Modern healthcare is shifting from traditional 2D diagnostic models to 3D systems such as:
- Organoids
- Spheroids
- Organ-on-chip technologies
These models enhance:
- Host-pathogen interaction studies
- Disease modeling
- Drug development
2. Growth of Point-of-Care Diagnostics
Point-of-care (POC) testing is gaining traction due to:
- Rapid results (within minutes)
- Decentralized testing capabilities
- Improved accessibility in remote areas
POC immunoassays are widely used for:
- Influenza
- Malaria
- HIV
- Tuberculosis
- COVID-19
Market Scope
| Report Coverage | Details |
|---|---|
| Market Size (2025) | USD 2.88 Billion |
| Market Size (2026) | USD 3.22 Billion |
| Market Size (2035) | USD 8.87 Billion |
| CAGR (2026–2035) | 11.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Forecast Period | 2026–2035 |
| Segments Covered | Product Type, Technology, Test Type, Disease Type, Application |
Market Dynamics
Drivers: Improved Detection Tools
- Rise of point-of-care testing
- Growing adoption of home-based diagnostic kits
- Integration with smartphones and cloud platforms
- Increasing demand for rapid test kits (results within 30 minutes)
Restraints: High Costs
- High cost of advanced analyzers and reagents
- Maintenance and calibration expenses
- Limited reimbursement policies in some regions
Opportunities: Integration with Advanced Technologies
- Use of nanotechnology (gold nanoparticles, quantum dots)
- Adoption of microfluidics systems
- Enhanced sensitivity and reduced testing time
Segmental Insights
Product Type Insights
- Reagents & Kits dominated (56.8% share in 2025) due to recurring demand
- Software & Data Management fastest-growing due to digital lab integration
Technology Insights
- Chemiluminescent Immunoassay (CLIA) led market due to high accuracy
- Multiplex Immunoassay expected fastest growth (13.4% CAGR)
Test Type Insights
- Antibody Detection Tests dominated market
- Combined Antigen & Antibody Tests growing rapidly for early detection
Disease Type Insights
- Hepatitis Immunoassays led with 23.8% share
- Respiratory Infection Immunoassays expected fastest growth
Application Insights
- Diagnostic Testing dominated (38.7%)
- Epidemiological Surveillance fastest-growing segment
Regional Insights
North America
- Largest market share (34.8%)
- Driven by:
- Advanced laboratory automation
- Strong government support
- Multiplex testing adoption
United States
- Strong growth due to:
- Favorable regulatory environment
- Advanced diagnostic infrastructure
- Clinical trial support systems
Asia-Pacific
- Fastest-growing region due to:
- Expanding healthcare infrastructure
- Rising elderly population
- Growing adoption of decentralized testing
India Market Trends
- Increasing preventive healthcare awareness
- Expansion of Bio-Safety Level 3 laboratories
- Strengthening of National Center for Disease Control (NCDC)
Leading Companies in the Market
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- bioMérieux SA
- Siemens Healthineers AG
- Beckman Coulter, Inc.
- QuidelOrtho Corporation
- Thermo Fisher Scientific Inc.
- Danaher Corporation
- Sysmex Corporation
- Becton, Dickinson and Company
- DiaSorin S.p.A.
- Fujirebio Holdings, Inc.
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
Recent Developments
- October 2025: Roche received CE mark for Elecsys Dengue Ag test, offering high specificity and sensitivity
- January 2025: bioMérieux acquired SpinChip Diagnostics to strengthen point-of-care immunoassay solutions
Market Segmentation
By Product Type
- Reagents & Kits
- Instruments & Analyzers
- Calibrators
- Controls
- Software & Data Management
By Technology
- Chemiluminescent Immunoassay
- ELISA
- Fluorescent Immunoassay
- Radioimmunoassay
- Rapid Lateral Flow Immunoassay
- Multiplex Immunoassay
By Test Type
- Antigen Detection
- Antibody Detection
- Combined Tests
- Confirmatory
- Quantitative
By Disease Type
- HIV
- Hepatitis
- Respiratory Infections
- TORCH
- STIs
- Gastrointestinal
- Vector-Borne Diseases
By Application
- Screening
- Diagnostic Testing
- Disease Monitoring
- Blood Screening
- Epidemiological Surveillance
Conclusion
The infectious disease immunoassay market is rapidly evolving with advancements in diagnostics, AI integration, and point-of-care technologies. Increasing global disease burden, demand for rapid testing, and healthcare modernization are key growth drivers.
With continuous innovation in multiplex assays, microfluidics, and digital diagnostics, the market is poised for robust expansion through 2035, offering significant opportunities for healthcare providers, diagnostic companies, and technology innovators.
Get Sample Copy: https://www.precedenceresearch.com/sample/8237
📩 For inquiries regarding discounts, bulk purchases, or customization requests, please contact:sales@precedenceresearch.com