Market Overview
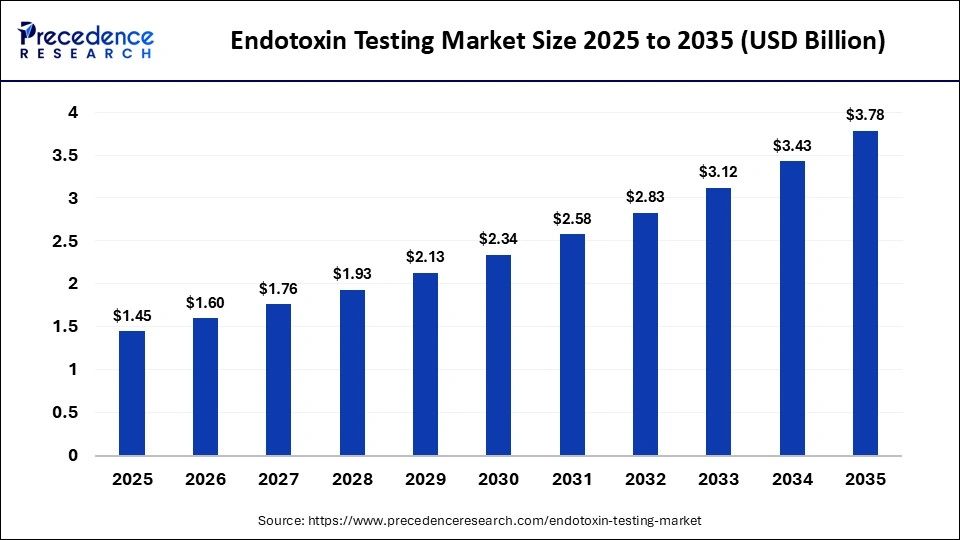
The global endotoxin testing market is experiencing strong growth due to increasing regulatory scrutiny and rising demand for safe pharmaceuticals and medical devices. The market size was valued at USD 1.45 billion in 2025 and is projected to grow from USD 1.60 billion in 2026 to approximately USD 3.78 billion by 2035, expanding at a CAGR of 10.05% during the forecast period.
This growth is primarily driven by the need to ensure sterility in injectable drugs, biologics, vaccines, and implantable medical devices. With stringent global regulatory standards and rising healthcare awareness, endotoxin testing has become a critical component of quality control in life sciences and healthcare manufacturing.
Read Also: AI for Scientific Discovery Market
Key Highlights
- Market Size 2026: USD 1.60 Billion
- Projected Market Size 2035: USD 3.78 Billion
- CAGR (2026–2035): 10.05%
- Leading Region: North America
- Fastest Growing Region: Asia Pacific
- Top Testing Method: Gel Clot Endotoxin Test
- Leading Application: Pharmaceutical Manufacturing
What is Endotoxin Testing?
Endotoxin testing refers to the detection and quantification of bacterial endotoxins—primarily lipopolysaccharides (LPS)—that can contaminate pharmaceutical products, biologics, and medical devices. These contaminants can cause severe immune reactions, making their detection essential for patient safety.
Testing is especially critical for:
- Injectable drugs
- Vaccines
- Implantable medical devices
- Biopharmaceuticals
With increasing production of biologics and sterile injectables, the demand for accurate and rapid endotoxin testing solutions continues to rise.
Market Drivers
1. Rising Demand for Safe Pharmaceuticals
The growing prevalence of chronic diseases has increased the use of injectable drugs and biologics, which require strict endotoxin control.
2. Stringent Regulatory Standards
Regulatory agencies such as the FDA and EMA mandate rigorous endotoxin testing, driving consistent demand for validated testing methods.
3. Growth in Biopharmaceutical Industry
The rapid expansion of biologics, biosimilars, and vaccines is significantly boosting the need for reliable endotoxin detection technologies.
4. Shift Toward Animal-Free Testing
The transition from traditional LAL (Limulus Amebocyte Lysate) tests to recombinant Factor C (rFC) methods is gaining momentum due to sustainability and ethical considerations.
Impact of AI on the Endotoxin Testing Market
Artificial Intelligence (AI) is revolutionizing endotoxin testing by enhancing speed, accuracy, and compliance. AI-driven systems enable:
- Automated data analysis and interpretation
- Real-time monitoring of contamination risks
- Predictive maintenance of testing equipment
- Reduction in false positives and human errors
Additionally, AI improves audit trails and ensures regulatory compliance by automating documentation and reporting processes, significantly enhancing laboratory efficiency.
Key Market Trends
Shift Toward Animal-Free Testing
There is a growing preference for recombinant technologies such as rFC and rCR, reducing reliance on horseshoe crab-derived LAL tests.
Automation and High-Throughput Systems
Laboratories are adopting automated platforms to increase testing efficiency and handle high sample volumes with minimal manual intervention.
Increasing Regulatory Pressure
Global regulatory bodies are enforcing stricter compliance, prompting manufacturers to invest in advanced endotoxin testing solutions.
Rise of Rapid and In-Process Testing
Real-time testing during production is gaining traction, allowing early detection of contamination and reducing product waste.
Segment Analysis
By Testing Method
The gel clot endotoxin test dominated the market in 2025 due to its:
- Cost-effectiveness
- Simplicity
- Strong regulatory acceptance
However, the chromogenic endotoxin test is expected to grow rapidly due to its ability to provide quantitative results, higher sensitivity, and compatibility with automation systems.
By Product Type
The endotoxin detection products segment held the largest share, driven by widespread use in pharmaceutical and biotechnology industries.
The PCR mycoplasma detection kits segment is projected to grow at the fastest rate due to:
- High sensitivity and accuracy
- Faster turnaround times
- Strong regulatory compliance
By Application
The pharmaceutical manufacturing segment led the market in 2025, supported by:
- Strict safety requirements for injectables
- High production of biologics and vaccines
- Regulatory mandates for quality control
The medical device manufacturing segment is expected to grow rapidly due to increasing demand for sterile and biocompatible devices.
Regional Insights
North America
North America dominated the market in 2025 due to:
- Strong regulatory frameworks
- Advanced pharmaceutical and biotech industries
- High adoption of automated testing technologies
The U.S. leads the region with widespread adoption of recombinant testing methods and strong FDA oversight.
Asia Pacific
Asia Pacific is the fastest-growing region, driven by:
- Expansion of biopharmaceutical manufacturing
- Rising demand for medical devices
- Increasing outsourcing of pharmaceutical production
Countries like India and China are emerging as key growth hubs due to large-scale production and improving regulatory compliance.
Europe
Europe is witnessing steady growth due to:
- Strong pharmaceutical manufacturing base
- Strict regulatory compliance
- Increasing investment in biologics and R&D
Countries such as Germany, France, and the UK are leading contributors to regional growth.
Competitive Landscape
The endotoxin testing market is highly competitive, with companies focusing on innovation, automation, and sustainable testing solutions.
Key Players Include:
- Charles River Laboratories International, Inc.
- Lonza Group Ltd.
- Merck KGaA
- Thermo Fisher Scientific Inc.
- bioMérieux SA
- Eurofins Scientific SE
- Sartorius AG
- Bio-Rad Laboratories, Inc.
- WuXi AppTec Co. Ltd.
- GenScript Biotech Corporation
Recent Developments
- January 2026: BETMAT Biotechnology launched sustainable endotoxin testing services using recombinant technologies (rFC and rCR).
- October 2024: LGM Pharma expanded analytical testing services to include rapid endotoxin testing.
- January 2024: Charles River Laboratories introduced Endosafe Trillium rCR cartridges for improved efficiency and sustainability.
Future Outlook
The endotoxin testing market is set for strong growth as the healthcare industry continues to prioritize safety and regulatory compliance. Future opportunities include:
- Wider adoption of recombinant and animal-free testing methods
- Integration of AI and automation in laboratories
- Expansion in emerging markets
- Growth in biologics and cell therapy production
As pharmaceutical and medical device industries evolve, endotoxin testing will remain a critical pillar in ensuring product safety and quality.
Conclusion
The global endotoxin testing market is on a steady growth trajectory, driven by stringent regulations, rising biologics production, and technological advancements. With a projected value of USD 3.78 billion by 2035, the market offers significant opportunities for innovation and expansion.
Companies investing in advanced, automated, and sustainable testing solutions will be well-positioned to capitalize on this growing demand.
Get Sample Copy: https://www.precedenceresearch.com/sample/7822
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com