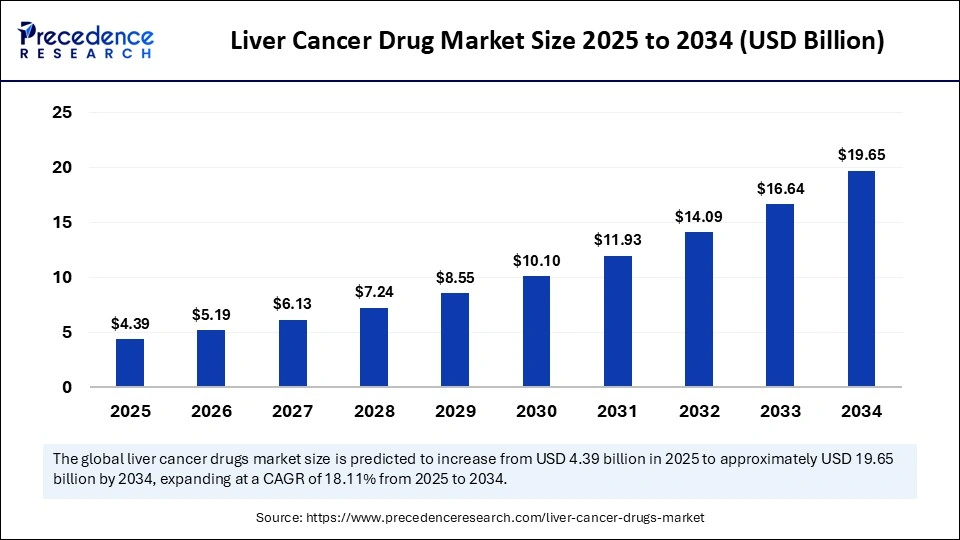
Market Highlights
- North America dominated the global liver cancer drug market with the largest share of 37.8% in 2024.
- The Asia Pacific is expected to grow at the highest CAGR of 12.4% from 2025 to 2034.
- By drug type, the targeted therapy segment contributed the largest market share of 41.8% in 2024.
- By drug type, the immunotherapy segment is growing at a notable CAGR of 11.6% from 2025 to 2034.
- By mechanism of action, the tyrosine kinase inhibitors segment led the market while holding the largest share of 46.4% in 2024.
- By mechanism of action, the checkpoint inhibitors segment is expected to grow at a 11.8% CAGR between 2025 and 2034.
- By formulation, the oral segment led the market while holding the largest share of 48.4% in 2024.
- By formulation, the injectable segment is growing at a strong CAGR of 11.7% between 2025 and 2034.
- By application, the advanced liver cancer segment led the market while holding the largest share of 51.5% in 2024.
- By application, the early-stage liver cancer segment is expected to grow at a 12.2% CAGR between 2025 and 2034.
- By end-user, the hospitals segment led the market while holding the largest share of 45.4% in 2024.
- By end-user, the specialty oncology clinics segment is expected to grow at a 12.0% CAGR between 2025 and 2034.
What is the Liver Cancer Drug Market?
The liver cancer drug market encompasses a range of pharmaceutical therapies developed to treat hepatocellular carcinoma (HCC) — the most common type of liver cancer — and other hepatic malignancies. The market is witnessing strong growth due to the rising global incidence of liver cancer, increasing investment in research and development (R&D), and rapid advancements in targeted and immunotherapeutic treatments.
Key treatment modalities include targeted therapies, immunotherapies, chemotherapy, and combination regimens, all designed to inhibit tumor progression, improve survival outcomes, and enhance patients’ quality of life. The introduction of immune checkpoint inhibitors and molecularly targeted drugs is transforming liver cancer care, allowing for more precise and effective therapeutic strategies.
The market is also benefiting from the growing adoption of personalized medicine, which tailors treatment based on a patient’s genetic and molecular profile. Increasing awareness of early diagnosis and government support for cancer research are further propelling demand for advanced therapies and early detection tools.
A recent milestone in this field was achieved in October 2025, when the Mayo Clinic became the first institution to administer an investigational radioactive therapy targeting glypican-3 (GPC3) — a protein overexpressed in HCC. This radiopharmaceutical theragnostic (RPT) therapy marks a breakthrough in precision oncology, representing the next frontier in targeted liver cancer treatment.
Key Technological Shifts in the Liver Cancer Drug Industry
The liver cancer drug industry is undergoing a major technological transformation, driven by the rapid adoption of precision medicine, targeted therapies, and AI-enabled drug discovery. This paradigm shift is reshaping the therapeutic landscape by focusing on molecularly guided treatments that inhibit key signaling pathways responsible for tumor growth and metastasis.
Leading targeted therapies such as sorafenib and lenvatinib have set the foundation for this evolution, offering improved disease management through pathway-specific inhibition. The next wave of innovation involves the strategic integration of targeted agents with immunotherapies and chemotherapeutic regimens, resulting in synergistic treatment outcomes and improved resistance management. These advancements are supported by rising R&D investments and adaptive clinical trial designs that enable the development of personalized treatment protocols tailored to individual genetic and molecular profiles.
In parallel, artificial intelligence (AI) and machine learning (ML) are transforming liver cancer research by accelerating drug discovery, biomarker identification, and treatment optimization. AI-powered platforms enhance the precision of target validation, reduce development timelines, and increase the success rate of clinical candidates.
Industry leaders such as Roche, Bristol-Myers Squibb, Bayer AG, Merck & Co., Exelixis, BeiGene, and Gilead Sciences are at the forefront of this transformation. These companies are investing in cutting-edge platforms including nanoparticle-based drug delivery, immuno-oncology, gene therapy, organ-on-a-chip models, and AI-driven radiomics to refine diagnosis and therapy.
Collaborative innovation is further accelerating progress — for instance, AI-integrated biomanufacturing platforms like Cellares’ automated cell therapy systems exemplify how technology is reshaping both discovery and production pipelines. Collectively, these advancements are propelling the liver cancer drug industry toward a future defined by personalized, data-driven, and highly effective cancer therapies.
Regional Insights
North America dominates the liver cancer drug market, primarily driven by the high incidence of hepatocellular carcinoma (HCC), robust healthcare infrastructure, and rapid adoption of advanced therapeutics. The United States holds the largest share, supported by strong R&D investments, frequent FDA approvals, and the presence of leading biopharmaceutical companies such as Bristol-Myers Squibb, Merck & Co., Gilead Sciences, and Exelixis. Increasing access to immunotherapies and targeted treatments, alongside government-backed cancer research programs, further fuels market growth.
Europe represents a significant market share, underpinned by widespread access to innovative oncology drugs and well-established clinical trial networks. Countries like Germany, the U.K., France, and Italy are at the forefront of adopting precision medicine and combination immunotherapies for liver cancer treatment. Supportive healthcare reimbursement systems, rising awareness of early diagnosis, and collaborative research initiatives across the EU Horizon programs contribute to sustained regional expansion.
The Asia-Pacific region is expected to witness the fastest growth in the liver cancer drug market, owing to its high disease burden, large patient population, and improving access to advanced cancer care. Countries such as China, Japan, South Korea, and India are experiencing a surge in liver cancer prevalence, largely linked to hepatitis B and C infections. Expanding healthcare infrastructure, government initiatives promoting early screening and vaccination, and increased clinical trial activity by global pharma firms are accelerating regional market development.
Latin America is emerging as a growing market due to improving access to cancer therapies and expanding public health initiatives. Brazil and Mexico lead the region, driven by the adoption of targeted therapies, increasing oncology-focused R&D, and growing partnerships between local health ministries and global pharmaceutical companies. However, limited reimbursement coverage and high treatment costs continue to pose challenges in some areas.
Recent Developments
- In August 2025, the study on the ImmunoTACE trial was published in Clinical Cancer Research, an academic journals, found enhanced progression-free survival (PFS) for patient with hepatocellular carcinoma (HCC). According to the study, patients who received the cell-based vaccine along with standard therapy saw a longer period of progression-free survival than patients who received standard therapy alone.
- In June 2025, UPMC Central PA Surgical Oncology introduced a specialized treatment, hepatic arterial infusion (HAI) pump therapy for patients with either metastatic cancer to the liver or cancers emerging in the liver. HAI therapy is able to more precisely target liver tumors to improve patient eligibility for curative-intent surgery and overall survival of the patients.
- In April 2025, the combination of nivolumab and ipilimumab received approval from the U.S. Food and Drug Administration as a prior treatment for adults with metastatic hepatocellular carcinoma. U.S. FDA has approved this therapy, due to positive results of the Phase 3 CheckMte 9DW trial, with significant improvement in survival with the immunotherapy-based combinations.
Get Sample Link : https://www.precedenceresearch.com/sample/7075
- Personalized Testing and Supplements Market Size to Surpass USD 7.74 Billion by 2034 - November 13, 2025
- Microinsurance Market Size to Surpass USD 161.54 Billion by 2034 - November 13, 2025
- Liver Cancer Drug Market Size to Surpass USD 19.65 Billion by 2034 - November 13, 2025
