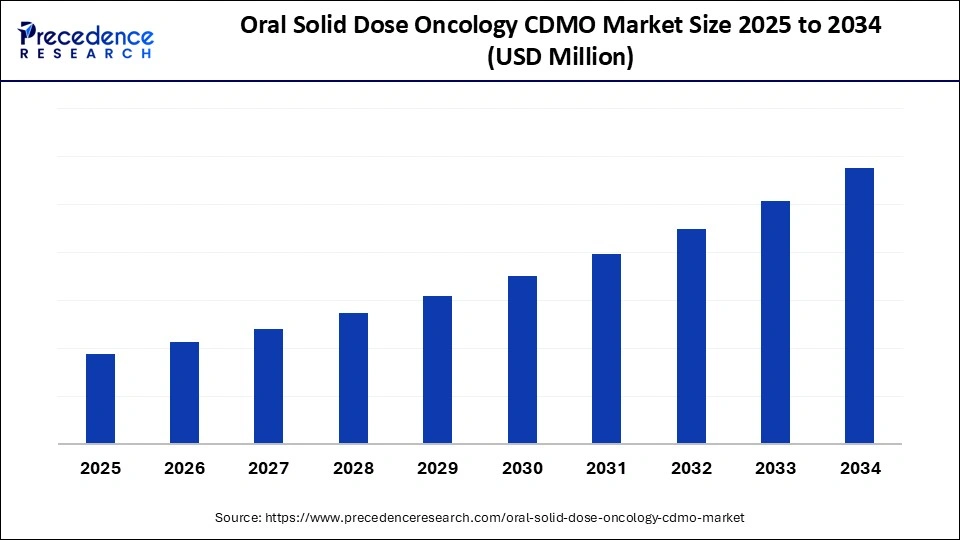
Oral Solid Dose Oncology CDMO Market Key Takeaways
- By region, North America dominated the market, holding the largest market share in 2024.
- By region, Asia Pacific is expected to expand at the fastest CAGR in the oral solid dose oncology CDMO market between 2025 and 2034.
- By service type, the commercial manufacturing & formulation/process development segment held the largest market share in 2024.
- By service type, high-potency is expected to grow at a remarkable CAGR between 2025 and 2034.
- By process type, the wet granulation & direct compression segments held the largest market share in 2024.
- By process type, advanced coating/modified-release tech and containment technologies for HPAPIs is expected to grow at a remarkable CAGR between 2025 and 2034.
- By product type, the small-molecule targeted oncology agents segment held the largest share in the oral solid dose oncology CDMO market during 2024.
- By product type, HPAPI handling and cytotoxic oral formulations are expected to grow at a remarkable CAGR between 2025 and 2034.
- By dosage form type, the immediate-release tablets segment held the largest market share in 2024.
- By dosage form type, modified release and multiarticulate systems are expected to grow at a remarkable CAGR between 2025 and 2034.
- By API potency type, the Non-HPAPI and standard small molecules segment held the largest market share in 2024.
- By API potency type, HPAPIs & cytotoxic containment services grow at a remarkable CAGR in the oral solid dose oncology CDMO market between 2025 and 2034.
- By customer type, the biotech & mid-sized pharma segment held the largest market share in 2024.
- By customer type, specialty/HPAPI-focused biotech is at a remarkable CAGR between 2025 and 2034.
- By contract type, the fee-for-service & turnkey contracts segment held the largest market share in 2024.
- By contract type, risk-sharing / milestone deals are at a remarkable CAGR between 2025 and 2034.
Market Overview
The Oral Solid Dose (OSD) Oncology CDMO Market encompasses contract development and manufacturing services dedicated to the formulation, scaling, production, testing, and packaging of oral solid oncology drugs such as tablets, capsules, and oral thin films. These services support both small-molecule oncology drugs and high-potency active pharmaceutical ingredients (HPAPIs), offering expertise in formulation design, process optimization, analytical testing, and regulatory compliance. CDMOs in this sector play a critical role across all stages of development—from clinical trial supply to commercial-scale production—enabling pharmaceutical innovators to bring new cancer therapies to market efficiently and safely.
Market growth is being propelled by a global transition toward patient-centered oncology care that emphasizes convenience, cost-effectiveness, and improved quality of life. Oral formulations have gained traction as a preferred mode of administration due to their ease of use, stability, and suitability for chronic cancer therapies, reducing dependence on hospital-based parenteral treatments. Advances in drug delivery technologies, including controlled-release mechanisms, solubility enhancement, and targeted oral delivery, are helping overcome traditional bioavailability barriers. Additionally, the increasing incidence of cancer, rising investment in personalized and home-based treatment regimens, and growing R&D activity in targeted oral therapies—such as kinase inhibitors and immunomodulators—are fueling demand for specialized CDMO partnerships. As pharmaceutical pipelines expand and regulatory expectations intensify, OSD oncology CDMOs are emerging as indispensable partners in driving innovation, scalability, and compliance across the oncology value chain.
Market Key Trends
- Rising Adoption of Targeted Oral Therapies and Immunomodulators: The oncology landscape is witnessing a rapid shift toward targeted oral therapies and immunomodulatory drugs that enable more precise and patient-friendly cancer treatment approaches.
- Surge in Personalized and Companion-Diagnostic-Linked Formulations: Increasing use of biomarkers and companion diagnostics is driving demand for personalized oral solid dose formulations tailored to individual patient profiles.
- Expansion in High-Potency API (HPAPI) Manufacturing Capabilities: CDMOs are investing heavily in containment technologies and specialized infrastructure to safely handle and manufacture high-potency oncology compounds.
- Integration of Digital Adherence Monitoring Tools: The incorporation of smart packaging and digital adherence tracking technologies supports home-based treatment regimens and improves therapeutic compliance.
- Growth in Strategic CDMO Partnerships: Pharmaceutical companies are expanding collaborations with CDMOs to accelerate development timelines, leverage specialized expertise, and enhance scalability in oncology drug production.
- Regulatory Encouragement for Novel OSD Oncology Approvals: Supportive regulatory pathways and expedited review processes are facilitating faster approval of innovative oral oncology therapies, encouraging greater investment in OSD development.
Market Driver
The Pill That Empowers: Patient-Centric Oncology
The primary growth driver of the oral solid dose (OSD) oncology CDMO market is its ability to empower patients through greater autonomy, comfort, and accessibility. Oral oncology therapies eliminate the need for frequent hospital visits, enabling patients to manage long-term treatment from home aligning perfectly with the global shift toward decentralized, patient-centric care. These formulations enhance adherence by simplifying administration and improving quality of life, particularly for chronic cancer patients. Pharmaceutical innovators are increasingly prioritizing oral formulations of kinase inhibitors, PARP inhibitors, and checkpoint modulators, reinforcing the trend toward self-managed cancer care. The growing acceptance of OSDs as a safe and effective alternative to parenteral therapies is driving demand for specialized CDMO capabilities, making patient convenience a defining force in the market’s expansion.
Market Restraint
The Bioavailability Bottleneck
Despite robust momentum, the OSD oncology CDMO market faces persistent challenges related to poor bioavailability and pharmacokinetic limitations of many anticancer molecules. Issues such as low solubility, poor permeability, and extensive first-pass metabolism often compromise therapeutic effectiveness. Moreover, developing oral formulations of high-potency compounds poses stringent safety and containment requirements, adding regulatory and operational complexity for CDMOs. Balancing potency, stability, and safety within tight formulation parameters remains a key scientific hurdle. Until advances in formulation science and harmonized regulatory frameworks fully address these challenges, the widespread adoption of complex oral oncology drugs will remain constrained.
Market Opportunity
Nanotech and Personalization: The Next Frontier
The most transformative opportunity in this market lies in integrating nanotechnology with personalized medicine to create next-generation oral oncology formulations. Nanocarriers such as liposomes, micelles, and polymeric nanoparticles are enabling the oral delivery of poorly soluble molecules, enhancing both absorption and targeted release. The fusion of genomic data and AI-driven formulation design opens the door to personalized dosing regimens that optimize therapeutic outcomes for each patient. Additionally, the rise of digital therapeutics and connected adherence tools promises to enhance real-world treatment success. Emerging markets—characterized by growing healthcare infrastructure, rising cancer incidence, and expanding access to affordable oncology care—offer vast untapped potential. Together, these innovations set the stage for a new era of smart, individualized, and accessible oral cancer therapies.
Regional Insights
Will North America Continue to Rule the Oral Solid Dose Oncology CDMO Market?
North America continues to dominate the oral solid dose (OSD) oncology CDMO market, supported by its advanced pharmaceutical ecosystem, extensive oncology R&D investments, and strong regulatory frameworks. The United States leads global innovation in targeted oral therapies—particularly kinase inhibitors, PARP inhibitors, and immunomodulators—benefiting from rapid clinical adoption, patent protection, and well-established reimbursement systems. The region’s CDMOs possess specialized expertise in high-potency API (HPAPI) manufacturing, containment technologies, and complex formulation development, aligning with the increasing demand for potent oral oncology drugs. Additionally, the growing emphasis on home-based cancer care and patient-centric treatment models is accelerating adoption of oral formulations. Robust venture funding, digital health integration, and advanced manufacturing capabilities ensure North America’s continued leadership in this space.
In Canada, healthcare modernization efforts and a growing number of precision oncology trials are driving interest in oral therapeutics. Canadian biotech firms are actively investing in nanotechnology-enabled OSD platforms to improve bioavailability and cost-effectiveness. Strategic cross-border collaborations with U.S. CDMOs are also streamlining technology transfer and accelerating commercialization timelines.
Why Is Asia-Pacific the Fastest Growing in the Oral Solid Dose Oncology CDMO Market?
The Asia-Pacific (APAC) region represents the fastest-growing market for OSD oncology CDMO services, propelled by a combination of rising cancer incidence, expanding healthcare access, and economic growth. Countries such as India, China, and Japan are investing heavily in local drug manufacturing to reduce dependency on imports and enhance affordability. Regional CDMOs are rapidly upgrading containment infrastructure and HPAPI-handling capabilities, positioning themselves as competitive global outsourcing hubs. Furthermore, government initiatives promoting pharmaceutical self-reliance, coupled with incentives for clinical research and technology transfer, are fostering a vibrant innovation ecosystem.
Rapid urbanization, an expanding middle-class population, and increasing awareness of oral cancer therapies are fueling market demand, while local academic–industry collaborations are catalyzing R&D in advanced oral delivery technologies and nanomedicine. With its blend of cost efficiency, skilled workforce, and growing technological sophistication, Asia-Pacific is poised to become the next global growth epicenter for oral oncology CDMO partnerships.
Recent Developments
- In September 2025, Oral liquid dosage forms play a crucial role in the pharmaceutical sector, particularly for patients who struggle with swallowing solid tablets, including children and the elderly. These liquid formulations facilitate more accurate dosing, which is especially advantageous in therapeutic areas like oncology. They also allow healthcare providers to modify dosages more easily to meet individual patient needs, thereby improving treatment effectiveness and adherence.
Get Sample Link : https://www.precedenceresearch.com/sample/7007
- Personalized Testing and Supplements Market Size to Surpass USD 7.74 Billion by 2034 - November 13, 2025
- Microinsurance Market Size to Surpass USD 161.54 Billion by 2034 - November 13, 2025
- Liver Cancer Drug Market Size to Surpass USD 19.65 Billion by 2034 - November 13, 2025
