The global rifaximin market is witnessing significant growth, driven by the rising prevalence of gastrointestinal disorders and the increasing demand for non-systemic antibiotics. According to market estimates, the global rifaximin market size was valued at USD 1.84 billion in 2024 and is projected to grow from USD 2.04 billion in 2025 to approximately USD 5.09 billion by 2034, registering a CAGR of 10.72% during the forecast period.
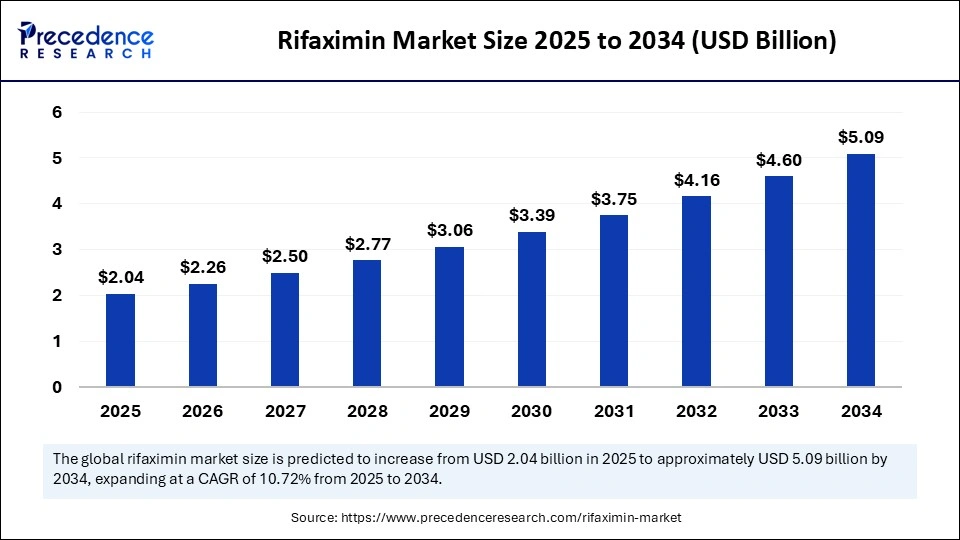
Table of Contents
ToggleRifaximin Market Key Takeaways
-
Market Size (2024): USD 1.84 billion
-
Market Size (2034): USD 5.09 billion
-
CAGR (2025–2034): 10.72%
-
Regional Leader: North America (40% share in 2024)
-
Fastest Growing Region: Asia Pacific
-
Leading Indication (2024): Irritable Bowel Syndrome with Diarrhea (IBS-D) – 45% share
-
Dosage Form: Tablets dominated with a 65% share in 2024
-
Distribution Channel: Retail pharmacies captured 50% market share
Market Overview
Rifaximin is a non-absorbable antibiotic primarily used for treating gastrointestinal disorders such as
-
Irritable bowel syndrome with diarrhea (IBS-D)
-
Hepatic encephalopathy (HE)
-
Traveler’s diarrhea
Unlike traditional antibiotics, rifaximin works locally in the gut without significant systemic absorption, reducing side effects while maintaining microbiota balance. Demand for rifaximin 550 mg tablets is especially strong due to its high efficacy in gastrointestinal treatments.
Regional Insights
North America
-
Held 40% market share in 2024, leading the global rifaximin market.
-
Growth is supported by advanced healthcare infrastructure and high patient awareness.
-
The U.S. market alone is projected to grow from USD 515.2 million in 2024 to USD 1.45 billion by 2034, expanding at a CAGR of 10.93%.
Asia Pacific
-
Expected to be the fastest-growing region from 2025 to 2034.
-
Growth drivers include rising disposable income, an aging population, and government investments in healthcare.
-
China and Japan are leading contributors, with strong regulatory frameworks and expanding pharmaceutical manufacturing capabilities.
Role of AI in Rifaximin Market
Artificial Intelligence is revolutionizing rifaximin manufacturing and supply chain processes
-
Polymorphic control & synthesis optimization → Better drug quality and efficiency.
-
Predictive analytics → Accurate reaction condition forecasts for production.
-
Automated quality control & regulatory submissions → Faster approvals and streamlined processes.
-
Supply chain management → Enhanced accessibility and reduced bottlenecks.
Market Drivers
-
Growing prevalence of gastrointestinal diseases worldwide.
-
Rising awareness about effective treatment options.
-
Strong demand for non-systemic antibiotics with minimal side effects.
-
Expanding insurance coverage and government healthcare initiatives.
-
Increased research into microbiome-focused treatments.
Market Restraints
-
Price fluctuations due to trade tariffs and supply chain challenges.
-
Patent expiry in 2028 for Xifaxan, leading to generic competition.
Opportunities
-
Technological and pharmaceutical innovations in rifaximin formulations.
-
Growing R&D in continuous manufacturing and nano-formulations.
-
Rising adoption of oral suspension forms among pediatric and elderly patients.
Recent Developments
-
June 2025 – Zydus Lifesciences received tentative FDA approval for generic rifaximin 550 mg tablets for IBS-D.
-
January 2025 – Bausch Health & Salix Pharmaceuticals announced CMS selection for Xifaxan (rifaximin) 550 mg under U.S. drug price negotiation programs.
Key Players in the Rifaximin Market
-
Dr. Reddy’s Laboratories
-
Sun Pharmaceutical Industries Ltd.
-
Lupin Limited
-
Cipla Ltd.
-
Pfizer Inc.
-
Novartis AG
-
Zydus Cadila
-
Teva Pharmaceutical Industries Ltd.
Segment Covered in the Report
By Indication
- Irritable Bowel Syndrome (IBS-D) with Diarrhea
- Adult Patients
- Pediatric Patients
- Hepatic Encephalopathy (HE)
- Prevention
- Treatment
- Traveler’s Diarrhea
- Prevention
- Treatment
- Others
- Small Intestinal Bacterial Overgrowth (SIBO)
- Infectious Diarrhea
By Dosage Form
- Tablets
- 200 mg
- 550 mg
- Oral Suspension
- 200 mg/5 mL
- Others
- Capsules
- Powder for Suspension
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
- Rifaximin Market Size to Hit USD 5.09 Billion by 2034 - August 29, 2025
- Ophthalmology PACS Market to Reach USD 333.32 Million by 2034 - August 29, 2025
- Behavioral Health Market to Surpass USD 330.35 Billion by 2034 - August 26, 2025
