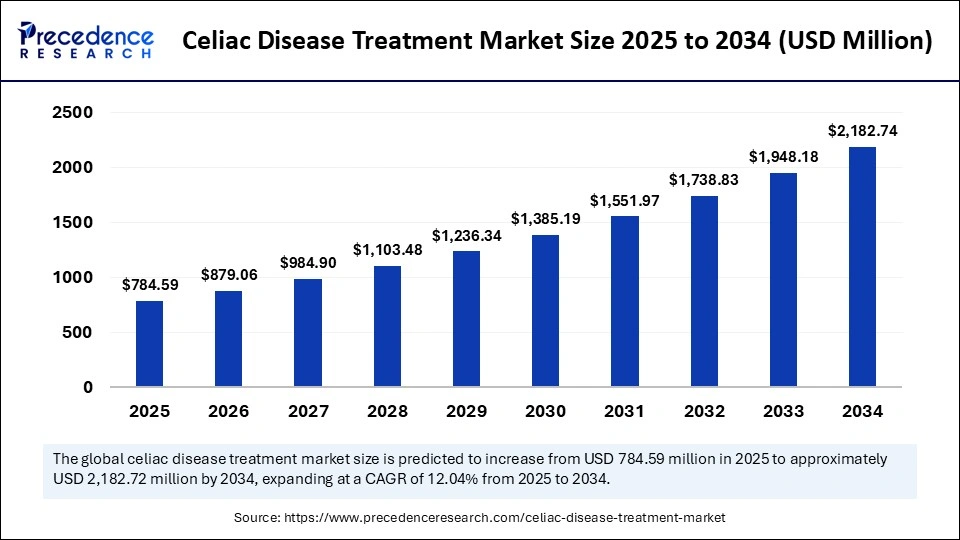
Market Highlights
- North America held the largest market share of 40% in 2024.
- The Asia Pacific is expected to experience the fastest CAGR from 2025-2034.
- By product type, the gluten-free drugs segment held the largest market share of 60% in 2024.
- By product type, the non-drug therapies segment is expected to witness the fastest growth from 2025-2034.
- By mechanism of action, the immune modulators segment contributed the highest market share of 40% in 2024.
- By mechanism of action, the enzyme therapy segment is expected to grow at the fastest CAGR from 2025-2034.
- By route of administration, the oral segment generated the biggest market share of 70% in 2024.
- By route of administration, the injectable segment is expected to grow at the fastest CAGR from 2025-2034.
- By end users, the hospitals & clinics segment accounted for the largest market share of 50% in 2024.
- By end users, the pharmacies & retail stores segment is expected to grow at the fastest CAGR from 2025-2034.
- By distribution channel, the hospital pharmacies segment held the largest market share of 45% in 2024.
- By distribution channel, the online/e-commerce segment is expected to grow at the fastest CAGR from 2025-2034.
What is the Celiac Disease Treatment Market?
The celiac disease treatment market encompasses a range of therapeutic solutions, pharmaceuticals, and management strategies designed to support individuals affected by celiac disease, an autoimmune disorder triggered by the ingestion of gluten. This market includes gluten-free dietary supplements, enzyme therapies, immunomodulatory drugs, and advanced diagnostic tools aimed at minimizing symptoms, preventing intestinal damage, and enhancing patient quality of life.
Market growth is primarily driven by the rising prevalence of celiac disease, improved diagnostic accuracy, and ongoing research into novel treatment approaches that go beyond strict gluten-free diets. Increasing awareness, coupled with biotechnological advancements and clinical trials for next-generation therapies, is further propelling the global celiac disease treatment market forward.
Key Technological Shifts in the Celiac Disease Treatment Market
The celiac disease treatment market is witnessing major technological advancements, with Artificial Intelligence (AI) emerging as a transformative force across diagnostics, drug discovery, and personalized patient care. AI-powered systems are revolutionizing disease detection by analyzing vast datasets of biopsy images and endoscopic scans to identify subtle histopathological features indicative of celiac disease with exceptional accuracy.
Recent studies reveal that AI-driven models, including deep learning algorithms, demonstrate higher diagnostic precision than human pathologists when interpreting video capsule endoscopy results a less invasive and increasingly preferred diagnostic technique. This breakthrough underscores AI’s growing role not only in early disease detection but also in accelerating drug discovery and tailoring treatment plans to individual patient profiles.
As digital health technologies continue to evolve, the integration of AI, machine learning, and advanced imaging analytics is expected to significantly enhance diagnostic speed, therapeutic development, and overall patient outcomes in the celiac disease treatment landscape.
Regional Insights
North America holds the dominant share of the celiac disease treatment market, driven by a high prevalence of diagnosed cases, growing public awareness, and robust healthcare infrastructure. The United States leads the region due to the increasing adoption of gluten-free diets, a strong presence of biopharmaceutical companies, and ongoing clinical research into novel therapies and diagnostic tools. Continuous government and non-profit initiatives promoting early screening and patient education further support market expansion.
Europe represents a significant share of the market, supported by a strong focus on celiac disease awareness, accurate diagnostics, and regulatory support for gluten-free food labeling. Countries such as the U.K., Germany, Italy, and Sweden have established advanced diagnostic frameworks and patient registries, promoting better disease management. The region’s high demand for innovative gluten-free formulations and immunotherapies also fuels the development of new treatment approaches across the continent.
The Asia-Pacific region is projected to be the fastest-growing market for celiac disease treatment, driven by rising diagnosis rates, an expanding middle-class population, and increasing awareness of gluten intolerance. Countries like India, China, Japan, and Australia are witnessing a growing number of diagnosed cases, supported by advancements in medical imaging, pathology, and AI-based diagnostic tools. The region’s evolving healthcare infrastructure and the increasing availability of gluten-free food alternatives present major growth opportunities for market players.
Latin America is experiencing steady growth in the celiac disease treatment market due to improving diagnostic capabilities, increased awareness campaigns, and the availability of affordable gluten-free products. Brazil and Mexico are leading markets in this region, where public health programs and patient advocacy groups are emphasizing the importance of early diagnosis and adherence to gluten-free diets.
Recent Developments
- In February 2025, AliveDx submitted a 510(k) premarket notification and is looking to gain approval from the U.S. FDA for its MosaiQ AiPlex Connective tissue diseases microarray, intended to enhance diagnostic precision and efficiency for the detection of celiac diseases.
- In September 2024, Barinthus Biotherapeutics initiated its first-in-human Phase 1 trial of VTP-1000 in adults having celiac disease. It’s a randomized, placebo-based clinical trial that consists of a controlled gluten challenge. It holds potential to evaluate the safety and tolerability of VTP-1000.
Get Sample Link : https://www.precedenceresearch.com/sample/7080
- Personalized Testing and Supplements Market Size to Surpass USD 7.74 Billion by 2034 - November 13, 2025
- Microinsurance Market Size to Surpass USD 161.54 Billion by 2034 - November 13, 2025
- Liver Cancer Drug Market Size to Surpass USD 19.65 Billion by 2034 - November 13, 2025
