Market Overview
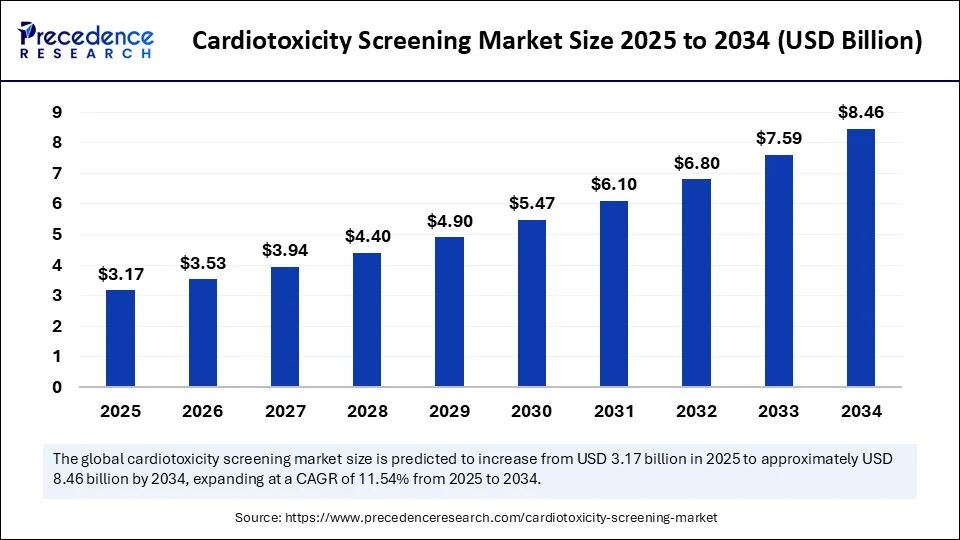
The cardiotoxicity screening market plays a vital role in ensuring the cardiac safety of pharmaceuticals and other chemical compounds. Cardiotoxicity screening encompasses a variety of approaches designed to detect adverse effects on heart function or structure early in drug development. With cardiovascular diseases being a leading cause of mortality worldwide, minimizing cardiotoxic side effects has become a priority. Regulatory mandates require thorough cardiotoxicity evaluation, making the cardiotoxicity screening market indispensable to pharmaceutical companies and research organizations focused on safety and efficacy.
AI and Innovation
Artificial intelligence and cutting-edge innovation are reshaping the cardiotoxicity screening market landscape. AI-based platforms enable predictive toxicology by analyzing complex biological datasets to forecast cardiotoxic risks with high precision. Innovations like 3D cardiac tissue models and microfluidic “heart-on-chip” devices simulate physiological conditions, enhancing screening reliability. Integration of AI with these experimental platforms accelerates drug screening and reduces false positives. Advances in bioinformatics and machine learning algorithms facilitate the identification of novel cardiotoxic biomarkers, thus enriching the cardiotoxicity screening market’s capabilities and efficiency.
Future Trends of Market
Future trends in the cardiotoxicity screening market include the expansion of personalized screening strategies using patient-derived cardiac cells to tailor drug safety assessments. The convergence of AI and big data analytics will refine predictive models, enabling earlier detection of cardiotoxicity. Real-time monitoring through wearable cardiac devices integrated with screening data could become a standard practice. Additionally, regulatory bodies are expected to endorse more predictive and less animal-dependent screening approaches. These developments promise to make cardiotoxicity screening more accurate, faster, and ethically aligned, driving market evolution.
Rising Demands of Market
The cardiotoxicity screening market is witnessing rising demand due to heightened regulatory requirements for comprehensive cardiac safety evaluation and the increasing complexity of novel drug molecules. Pharmaceutical pipelines are expanding with biologics and targeted therapies, some of which may carry unforeseen cardiac risks. Growing investment in cardiovascular disease research and awareness about drug-induced heart injuries among healthcare professionals amplify the market’s importance. Furthermore, advancements in diagnostic technology enable earlier detection of cardiotoxic effects, fueling the need for sophisticated screening tools.
Key Market Highlights
Notable market highlights include the growing adoption of high-throughput and high-content screening technologies that significantly reduce the time and cost associated with cardiotoxicity testing. The launch of innovative biomarker panels and imaging modalities has improved the accuracy of cardiac risk assessment. Collaborations between pharmaceutical companies and technology providers have resulted in the development of integrated screening platforms. Regulatory emphasis on early detection and risk mitigation continues to drive investments in cardiotoxicity screening. The ongoing evolution of standards for cardiac safety testing marks a critical milestone for the market.
Market Growth Drivers
Growth drivers for the cardiotoxicity screening market include increasing incidence of drug-induced cardiac complications and the need to minimize costly late-stage drug failures. Regulatory agencies worldwide are enforcing stricter guidelines to ensure cardiac safety, compelling drug developers to adopt advanced screening techniques. Rising healthcare expenditures and expanding pharmaceutical R&D budgets support the market’s growth. The increasing adoption of novel technologies like iPSC-derived cardiomyocytes and organ-on-chip systems enhances screening accuracy, thereby driving the market forward. Collaborations and funding for cardiovascular research also underpin growth momentum.
Restraints
Several factors restrain the cardiotoxicity screening market, including the high capital investment required for sophisticated screening platforms, which may be prohibitive for smaller companies. Scientific challenges related to replicating human cardiac physiology in vitro can lead to variable results.
Regulatory uncertainty concerning the validation and acceptance of novel screening methods may delay adoption. Additionally, limited availability of qualified personnel and standardized protocols can impede widespread implementation. These hurdles need to be addressed to unlock the full potential of the cardiotoxicity screening market.
Opportunities
The cardiotoxicity screening market offers promising opportunities through the expansion into emerging pharmaceutical hubs with increasing R&D activities. Development of cost-effective and automated screening technologies can enhance accessibility. There is also scope for diversification into non-pharmaceutical sectors such as chemical safety testing and environmental monitoring.
Advances in AI and data analytics present opportunities to develop predictive models that can be licensed or integrated into existing drug development pipelines. Partnerships between academic institutions and industry can foster innovation and accelerate the commercialization of new cardiotoxicity screening solutions
Also Read@ https://www.pharma-geek.com/dehydration-monitoring-devices-market/
Cardiotoxicity Screening Market Companies

- Charles River Laboratories
- Labcorp/Covance
- Eurofins Scientific
- WuXi AppTec
- Evotec
- ICON plc
- Certara
- Simulations Plus
- Axion BioSystems
- Multi-Channel Systems (MCS)
- Nanion Technologies
- Sophion Bioscience
- Molecular Devices/Danaher group
- Fujifilm-CDI (Cellular Dynamics International)
- Ncardia (formerly Axiogenesis)
- Axol Bioscience
- Thermo Fisher Scientific
- PerkinElmer
- Charles River/HESI/CiPA collaborators
Recent Developments
- In June 2025, the U.S. Geological Survey published in the journal Environmental Science and Technology, identifying Biosphenol P (BPP) as a potent cardiotoxic compound through high-throughput screening. Researchers have highlighted BPP’s ability to induce cardiotoxicity by apoptosis and the NF-kB pathway.
- In April 2025, the industry-leading solutions to streamline high-throughput screening while maintaining physiological relevance were launched by the Ncardia in a “Advancing high-throughput cardiac screening with physiologically relevant 3D iPSC-based models,” focused webinar.
Get Free sample link @ https://www.precedenceresearch.com/sample/6624
- Bioinformatics-as-a-Service (BaaS) Market Size to Reach USD 14.56 Billion by 2034 - October 8, 2025
- Exosome Therapeutics Market Size to Reach USD 4,020.75 Thousand by 2034 - October 7, 2025
- Microalgae Biotechnology Market Size, Report by 2034 - October 7, 2025
